Understanding the Basics of NH4OH: All About Ammonium Hydroxide
Introduction:
Welcome to the fascinating world of chemistry! Today, we are going to learn about NH4OH, also known as ammonium hydroxide. This might sound complicated, but don’t worry. We will explain everything step by step, making it easy and fun to understand. So, let’s dive in and discover what NH4OH is and why it’s important baddies west.
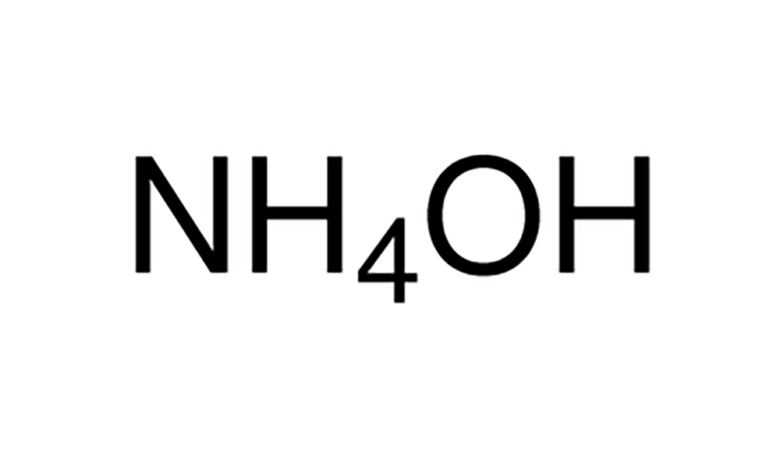
What is NH4OH (Ammonium Hydroxide)?
First, let’s understand what NH4OH is. It is a special chemical compound, made when ammonia (NH3) is dissolved in water (H2O). However, this mixture is called ammonium hydroxide. It has different names like ammonia water, ammonia liquor, and aqueous ammonia. However, you can think of it as a mixture that has ammonia gas in water.
Basicity of NH4OH:
So, when ammonia is mixed with water, something interesting happens. The water gives a part of itself called a proton to the ammonia. However, this creates two new parts: an ammonium cation (NH4+) and a hydroxide ion (OH–).
Saturated Solutions of Ammonium Hydroxide:
Next, let’s talk about saturated solutions. Finally, when we dissolve a lot of ammonia in water, we get a saturated solution of ammonium hydroxide. However, the amount of ammonia that can dissolve decreases if the water gets warmer. For example, if the water is at a certain temperature. However the solution might contain 35.6% ammonia by weight. This means that for every liter of solution, there are 308 grams of ammonia.
Read Also: ibommahub
Properties of Ammonium Hydroxide (NH4OH):
Now, let’s look at some important properties of it:
- Molar Mass: The molar mass of NH4OH is 35.04 grams per mole.
- Appearance: It is a colorless liquid.
- Smell: It has a strong, fishy odor.
- Density: The density can vary, but for a 25% solution, it’s about 0.91 grams per cubic centimeter.
- Melting and Boiling Points: A 25% solution melts at 215.7 K and boils at 310.8 K.
- Enthalpy of Formation: Its standard enthalpy of formation is -80 Kilojoules per mole.
Uses of Ammonium Hydroxide:
It is very useful and is used in many ways:
- Fertilizers: It is used to make chemical fertilizers, helping plants grow better.
- Chemical Manufacturing: It helps make chemicals that contain nitrogen, like nitric acid.
- Wood Treatment: It gives wood a dark, stained look, often used in furniture making.
Safety and Hazards
While it is useful, it can also be dangerous:
- Skin Contact: It can cause burns and irritation.
- Eye Contact: It can hurt the eyes and cause swelling.
- Inhalation: Breathing in the mist can damage the lungs and throat.
Safety Tips
- Wash with Water: If NH4OH touches your skin, wash it off with lots of water.
- Ventilation: Make sure to use it in a well-ventilated area.
ConclUnderstanding the Basics of NH4OH: All About Ammonium Hydroxideusion:
In conclusion, NH4OH, or ammonium hydroxide, is an important chemical with many uses. It’s a mixture of ammonia and water, and however it helps make fertilizers, cleaning agents, and more. However, it can be dangerous, so it’s important to handle it safely. Now you know all about NH4OH and why it’s important!
However by understanding the properties, its uses, and safety measures of NH4OH, we can appreciate its role in our daily lives and in various industries. Remember to always handle chemicals with care and stay safe!
Frequently Asked Questions (FAQs):
What is NH4OH in chemistry?
NH4OH, or ammonium hydroxide, is a solution of ammonia in water.
Is ammonium hydroxide harmful to humans?
Yes, it can be harmful. However, It can cause skin and eye irritation, burns, and respiratory issues if inhaled.
What is the difference between ammonia and ammonium hydroxide?
Ammonia is a gas (NH3), while ammonium hydroxide is ammonia dissolved in water (NH4OH).
Is ammonium hydroxide corrosive?
Yes, it is corrosive and can cause damage to skin, eyes, and lungs.
What pH is ammonia solution?
Ammonia solution has a pH of around 11, making it basic.
How strong is ammonium hydroxide?
The strength of ammonium hydroxide can vary, with solutions ranging from less than 1% to about 35% ammonia by concentration.
Stay safe and enjoy learning about the amazing world of chemistry!



